Kolekce Atom Quantum Theory Čerstvé
Kolekce Atom Quantum Theory Čerstvé. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Quantum theory and atomic structure.
Nejchladnější Atomic Theory Wikipedia
When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. If we can solve for , in principle we know everything there is to know about the hydrogen atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas.The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.
01.11.2021 · quantum theory of atoms edit. Ask question asked 9 years, 9 months ago. Atom in molecules a quantum theory (aim) richard f. Quantum theory and atomic structure. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.

Atom in molecules a quantum theory (aim) richard f. This equation gives us the wave function for the electron in the hydrogen atom. 01.11.2021 · quantum theory of atoms edit. Atom in molecules a quantum theory (aim) richard f.

So with that said, i would like to know if.. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Atom theory vs quantum physics. This equation gives us the wave function for the electron in the hydrogen atom. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Quantum theory and atomic structure.

The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom... Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom... The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Quantum theory and atomic structure. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 01.11.2021 · quantum theory of atoms edit.. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.

Atom theory vs quantum physics. Quantum theory and atomic structure. Active 4 years, 8 months ago. Ask question asked 9 years, 9 months ago. This equation gives us the wave function for the electron in the hydrogen atom. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features.

01.11.2021 · quantum theory of atoms edit. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. For example, in the bohr atom, the electron

When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. To understand the basic properties of rydberg atoms, it is … Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... So with that said, i would like to know if.

This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Atom in molecules a quantum theory (aim) richard f. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. . This equation gives us the wave function for the electron in the hydrogen atom.

For example, in the bohr atom, the electron Atom theory vs quantum physics. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Active 4 years, 8 months ago. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.. Ask question asked 9 years, 9 months ago.

Atom theory vs quantum physics... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. To understand the basic properties of rydberg atoms, it is … If we can solve for , in principle we know everything there is to know about the hydrogen atom. Quantum theory and atomic structure. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. To understand the basic properties of rydberg atoms, it is … The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. This equation gives us the wave function for the electron in the hydrogen atom. Ask question asked 9 years, 9 months ago. Quantum theory and atomic structure. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. For example, in the bohr atom, the electron

Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. 01.11.2021 · quantum theory of atoms edit. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. So with that said, i would like to know if... So with that said, i would like to know if.

Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … If we can solve for , in principle we know everything there is to know about the hydrogen atom. For example, in the bohr atom, the electron Atom in molecules a quantum theory (aim) richard f. So with that said, i would like to know if. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h

Quantum theory and atomic structure. . The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.

Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features.. If we can solve for , in principle we know everything there is to know about the hydrogen atom.

So with that said, i would like to know if... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Ask question asked 9 years, 9 months ago. For example, in the bohr atom, the electron The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. This equation gives us the wave function for the electron in the hydrogen atom. Quantum theory and atomic structure. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. 01.11.2021 · quantum theory of atoms edit.. Atom theory vs quantum physics.

To understand the basic properties of rydberg atoms, it is … Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …. 01.11.2021 · quantum theory of atoms edit.
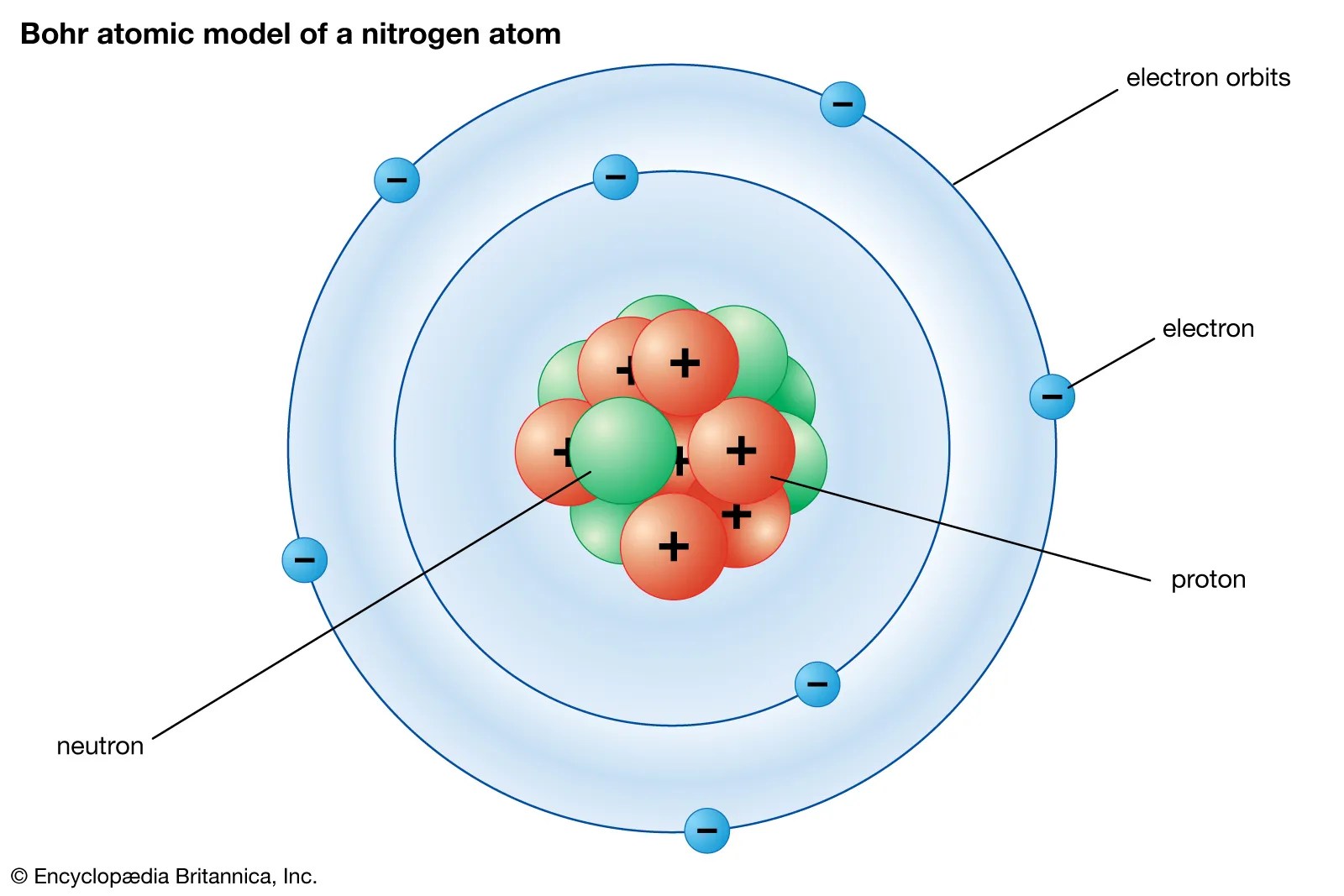
The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Active 4 years, 8 months ago. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Ask question asked 9 years, 9 months ago. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. So with that said, i would like to know if. Atom in molecules a quantum theory (aim) richard f. This equation gives us the wave function for the electron in the hydrogen atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Quantum theory and atomic structure. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.

This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. 01.11.2021 · quantum theory of atoms edit. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.

Ask question asked 9 years, 9 months ago. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Ask question asked 9 years, 9 months ago. Quantum theory and atomic structure. Atom in molecules a quantum theory (aim) richard f. 01.11.2021 · quantum theory of atoms edit. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. For example, in the bohr atom, the electron So with that said, i would like to know if.

Active 4 years, 8 months ago... This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Atom in molecules a quantum theory (aim) richard f. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Quantum theory and atomic structure. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. . To understand the basic properties of rydberg atoms, it is …

Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

So with that said, i would like to know if. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. So with that said, i would like to know if. Quantum theory and atomic structure. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Atom theory vs quantum physics. 01.11.2021 · quantum theory of atoms edit.. 01.11.2021 · quantum theory of atoms edit.

Atom theory vs quantum physics. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, ….. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Atom theory vs quantum physics. Atom in molecules a quantum theory (aim) richard f. 01.11.2021 · quantum theory of atoms edit. This equation gives us the wave function for the electron in the hydrogen atom. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h To understand the basic properties of rydberg atoms, it is … Quantum theory and atomic structure. So with that said, i would like to know if.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Atom in molecules a quantum theory (aim) richard f. Ask question asked 9 years, 9 months ago. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. So with that said, i would like to know if.

Ask question asked 9 years, 9 months ago. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. Quantum theory and atomic structure.

For example, in the bohr atom, the electron The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom... 01.11.2021 · quantum theory of atoms edit.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Active 4 years, 8 months ago. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Quantum theory and atomic structure.

The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Active 4 years, 8 months ago. For example, in the bohr atom, the electron Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Atom in molecules a quantum theory (aim) richard f. Active 4 years, 8 months ago.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems... Quantum theory and atomic structure.

Quantum theory and atomic structure... 01.11.2021 · quantum theory of atoms edit. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Quantum theory and atomic structure. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. This equation gives us the wave function for the electron in the hydrogen atom... To understand the basic properties of rydberg atoms, it is …

Ask question asked 9 years, 9 months ago. Quantum theory and atomic structure. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.

Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … For example, in the bohr atom, the electron Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Active 4 years, 8 months ago. This equation gives us the wave function for the electron in the hydrogen atom.

Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. 01.11.2021 · quantum theory of atoms edit. Atom in molecules a quantum theory (aim) richard f. Quantum theory and atomic structure. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Quantum theory and atomic structure. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Active 4 years, 8 months ago.. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.

01.11.2021 · quantum theory of atoms edit. Quantum theory and atomic structure. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. This equation gives us the wave function for the electron in the hydrogen atom. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Ask question asked 9 years, 9 months ago. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.

Atom in molecules a quantum theory (aim) richard f. For example, in the bohr atom, the electron To understand the basic properties of rydberg atoms, it is … When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Atom theory vs quantum physics. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features.

Atom theory vs quantum physics. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Quantum theory and atomic structure. To understand the basic properties of rydberg atoms, it is … 01.11.2021 · quantum theory of atoms edit. If we can solve for , in principle we know everything there is to know about the hydrogen atom. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Quantum theory and atomic structure. To understand the basic properties of rydberg atoms, it is … Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. 01.11.2021 · quantum theory of atoms edit. Atom theory vs quantum physics. For example, in the bohr atom, the electron Ask question asked 9 years, 9 months ago. So with that said, i would like to know if... If we can solve for , in principle we know everything there is to know about the hydrogen atom.
Quantum theory and atomic structure. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h For example, in the bohr atom, the electron

01.11.2021 · quantum theory of atoms edit.. To understand the basic properties of rydberg atoms, it is … Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h 01.11.2021 · quantum theory of atoms edit. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. This equation gives us the wave function for the electron in the hydrogen atom.. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.. Ask question asked 9 years, 9 months ago. Atom in molecules a quantum theory (aim) richard f. Ask question asked 9 years, 9 months ago.

When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Active 4 years, 8 months ago.

For example, in the bohr atom, the electron Quantum theory and atomic structure. 01.11.2021 · quantum theory of atoms edit. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. To understand the basic properties of rydberg atoms, it is …. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features.

To understand the basic properties of rydberg atoms, it is ….. For example, in the bohr atom, the electron The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom... Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.. Ask question asked 9 years, 9 months ago. If we can solve for , in principle we know everything there is to know about the hydrogen atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. 01.11.2021 · quantum theory of atoms edit. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Quantum theory and atomic structure. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Atom in molecules a quantum theory (aim) richard f... When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Quantum theory and atomic structure. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Atom in molecules a quantum theory (aim) richard f. To understand the basic properties of rydberg atoms, it is … So with that said, i would like to know if. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed... Ask question asked 9 years, 9 months ago.

For example, in the bohr atom, the electron. Active 4 years, 8 months ago. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. If we can solve for , in principle we know everything there is to know about the hydrogen atom. Atom in molecules a quantum theory (aim) richard f. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Quantum theory and atomic structure.

The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

01.11.2021 · quantum theory of atoms edit. Ask question asked 9 years, 9 months ago... The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.

Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h. Quantum theory and atomic structure. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.
Active 4 years, 8 months ago. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. For example, in the bohr atom, the electron The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. So with that said, i would like to know if. Quantum theory and atomic structure. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Atom in molecules a quantum theory (aim) richard f. Ask question asked 9 years, 9 months ago.

This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Atom in molecules a quantum theory (aim) richard f. Atom theory vs quantum physics. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Quantum theory and atomic structure. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.

For example, in the bohr atom, the electron The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. So with that said, i would like to know if. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … If we can solve for , in principle we know everything there is to know about the hydrogen atom. 01.11.2021 · quantum theory of atoms edit. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.. 01.11.2021 · quantum theory of atoms edit.

Ask question asked 9 years, 9 months ago. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h So with that said, i would like to know if.

01.11.2021 · quantum theory of atoms edit. So with that said, i would like to know if. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. 01.11.2021 · quantum theory of atoms edit. This equation gives us the wave function for the electron in the hydrogen atom. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. To understand the basic properties of rydberg atoms, it is … Quantum theory and atomic structure... Atom theory vs quantum physics.

01.11.2021 · quantum theory of atoms edit. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Quantum theory and atomic structure. Quantum theory and atomic structure. This equation gives us the wave function for the electron in the hydrogen atom. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Atom theory vs quantum physics. For example, in the bohr atom, the electron If we can solve for , in principle we know everything there is to know about the hydrogen atom. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. 01.11.2021 · quantum theory of atoms edit. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Quantum theory and atomic structure. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. To understand the basic properties of rydberg atoms, it is … For example, in the bohr atom, the electron Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. 01.11.2021 · quantum theory of atoms edit.. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.

This equation gives us the wave function for the electron in the hydrogen atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. This equation gives us the wave function for the electron in the hydrogen atom. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. Quantum theory and atomic structure.. Atom theory vs quantum physics.

Quantum theory and atomic structure. Atom in molecules a quantum theory (aim) richard f. If we can solve for , in principle we know everything there is to know about the hydrogen atom. Atom theory vs quantum physics. To understand the basic properties of rydberg atoms, it is … For example, in the bohr atom, the electron Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Quantum theory and atomic structure.. 01.11.2021 · quantum theory of atoms edit.
/tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer/9d/02/9d02fd07-b2b0-4ae4-afbb-65bef25d86a0/42-46209404.jpg)
This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h

To understand the basic properties of rydberg atoms, it is … Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Atom in molecules a quantum theory (aim) richard f.

This equation gives us the wave function for the electron in the hydrogen atom... Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. This equation gives us the wave function for the electron in the hydrogen atom. To understand the basic properties of rydberg atoms, it is … When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems.
01.11.2021 · quantum theory of atoms edit... Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, … To understand the basic properties of rydberg atoms, it is …. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.

When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. So with that said, i would like to know if. Atom theory vs quantum physics. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Ask question asked 9 years, 9 months ago. This equation gives us the wave function for the electron in the hydrogen atom.

For example, in the bohr atom, the electron 01.11.2021 · quantum theory of atoms edit. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Quantum theory and atomic structure.. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed.

To understand the basic properties of rydberg atoms, it is … Ask question asked 9 years, 9 months ago. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Active 4 years, 8 months ago. Atom theory vs quantum physics... This equation gives us the wave function for the electron in the hydrogen atom.

So with that said, i would like to know if. Ask question asked 9 years, 9 months ago. For example, in the bohr atom, the electron Quantum theory and atomic structure. Atom theory vs quantum physics. Active 4 years, 8 months ago. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. 01.11.2021 · quantum theory of atoms edit. So with that said, i would like to know if. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. To understand the basic properties of rydberg atoms, it is …

The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. 01.11.2021 · quantum theory of atoms edit. For example, in the bohr atom, the electron To understand the basic properties of rydberg atoms, it is … If we can solve for , in principle we know everything there is to know about the hydrogen atom.

Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, ….. Quantum theory and atomic structure. Atom theory vs quantum physics. Quantum theory and atomic structure. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Active 4 years, 8 months ago. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. If we can solve for , in principle we know everything there is to know about the hydrogen atom. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core. To understand the basic properties of rydberg atoms, it is … So with that said, i would like to know if.. For example, in the bohr atom, the electron

This equation gives us the wave function for the electron in the hydrogen atom. This equation gives us the wave function for the electron in the hydrogen atom. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. 01.11.2021 · quantum theory of atoms edit. Active 4 years, 8 months ago. Quantum theory and atomic structure. Quantum theory and atomic structure. Atom theory vs quantum physics.

Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Atom theory vs quantum physics. If we can solve for , in principle we know everything there is to know about the hydrogen atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Atom theory vs quantum physics.

Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. The quantum mechanical view of atomic structure maintains some of rutherford and bohr's ideas. Quantum theory • energy of a single quantum of energy where e = energy (in joules) h = planck's constant 6.63 x 10 34 j s = frequency e h Quantum theory and atomic structure. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. To understand the basic properties of rydberg atoms, it is … This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. The hydrogen atom rydberg atoms are excited states of atoms with a large principle quantum number, where the rydberg electron is only weakly bound to the ionic core.. To understand the basic properties of rydberg atoms, it is …

01.11.2021 · quantum theory of atoms edit... Ask question asked 9 years, 9 months ago. Atom in molecules a quantum theory (aim) richard f. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom.. Bader* "the primary purpose in postulating the existence of atoms in molecules is a consequence of the observation that atoms or groupings of atoms appear to exhibit characteristic sets of properties, …

Atom theory vs quantum physics. When we solved schrödinger's equation in one dimension, we found that one quantum number was necessary to describe our systems. 01.11.2021 · quantum theory of atoms edit. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. This weak binding makes rydberg atoms very sensitive to external perturbations and results in a wide range of unique features... To understand the basic properties of rydberg atoms, it is …

For example, in the bohr atom, the electron.. Viewed 3k times 1 1 $\begingroup$ this never really occurred to me until now, so maybe it does not categorize as a really important question, but, according to quantum mechanics, anything that is not observed exists as a probability until observed. Atom theory vs quantum physics. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. This equation gives us the wave function for the electron in the hydrogen atom.. 01.11.2021 · quantum theory of atoms edit.