Seznamy 167 Atom 3 Subatomic Particles Vynikající
Seznamy 167 Atom 3 Subatomic Particles Vynikající. What subatomic particles make up an atom and atoms? Subatomic particles are those which make up an atom.they are: It is neither negative nor positive.
Tady Solved 3 A Name The Three Subatomic Particles Which Chegg Com
It consists of three fundamental particles known as quarks. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. It is neither negative nor positive. 06.12.2011 · what are 3 subatomic particles? The electrons (or more specifically, electron clouds) orbit a small.22.08.2017 · there are basically three subatomic particles inside an atom.
It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. Subatomic particle relative mass relative charge; It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. It is neither negative nor positive. 27.04.2020 · particles that are smaller than the atom are called subatomic particles.

22.08.2017 · there are basically three subatomic particles inside an atom. Subatomic particles are those which make up an atom.they are: 27.03.2021 · a typical atom consists of three subatomic particles: Subatomic particle relative mass relative charge;.. Atoms are made up of particles called protons, neutrons, and electrons, …

It is the neutral part of an atom... Subatomic particle relative mass relative charge; First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … It is the neutral part of an atom.
Atoms are made up of particles called protons, neutrons, and electrons, … 27.03.2021 · a typical atom consists of three subatomic particles: It is the neutral part of an atom. Subatomic particles are those which make up an atom.they are: If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. Protons and neutrons are the subatomic particles that …. It is the neutral part of an atom.

The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. The center of the atom is called the nucleus. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Subatomic particle relative mass relative charge; It is the neutral part of an atom. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … The subatomic particles are the particles that comprise each atom. 22.08.2017 · there are basically three subatomic particles inside an atom. Protons and neutrons are the subatomic particles that …. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization.
.PNG)
The three main subatomic particles that form an atom are protons, neutrons, and electrons. It consists of three fundamental particles known as quarks. The electrons (or more specifically, electron clouds) orbit a small. Protons, neutrons, and electrons (as seen in the helium atom below). Protons and neutrons are the subatomic particles that … Subatomic particles are those which make up an atom.they are: It is neither negative nor positive. The three main subatomic particles that form an atom are protons, neutrons, and electrons. What subatomic particles make up an atom and atoms?.. 22.08.2017 · there are basically three subatomic particles inside an atom.
Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. 27.04.2020 · particles that are smaller than the atom are called subatomic particles... 27.03.2021 · a typical atom consists of three subatomic particles:

First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … It means that it has no charge. The center of the atom is called the nucleus. Protons and neutrons are the subatomic particles that … It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a …. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

What subatomic particles make up an atom and atoms? It means that it has no charge.
Subatomic particle relative mass relative charge; 06.12.2011 · what are 3 subatomic particles? The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. Protons, neutrons, and electrons (as seen in the helium atom below)... It is neither negative nor positive.

It is situated in the ….. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … 27.04.2020 · particles that are smaller than the atom are called subatomic particles. The electrons (or more specifically, electron clouds) orbit a small.. Atoms are made up of particles called protons, neutrons, and electrons, …

It means that it has no charge. The center of the atom is called the nucleus. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 22.08.2017 · there are basically three subatomic particles inside an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. All three subatomic particles have different subatomic particles mass. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. It is neither negative nor positive. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. Protons and neutrons are the subatomic particles that … All three subatomic particles have different subatomic particles mass.. It is the neutral part of an atom.
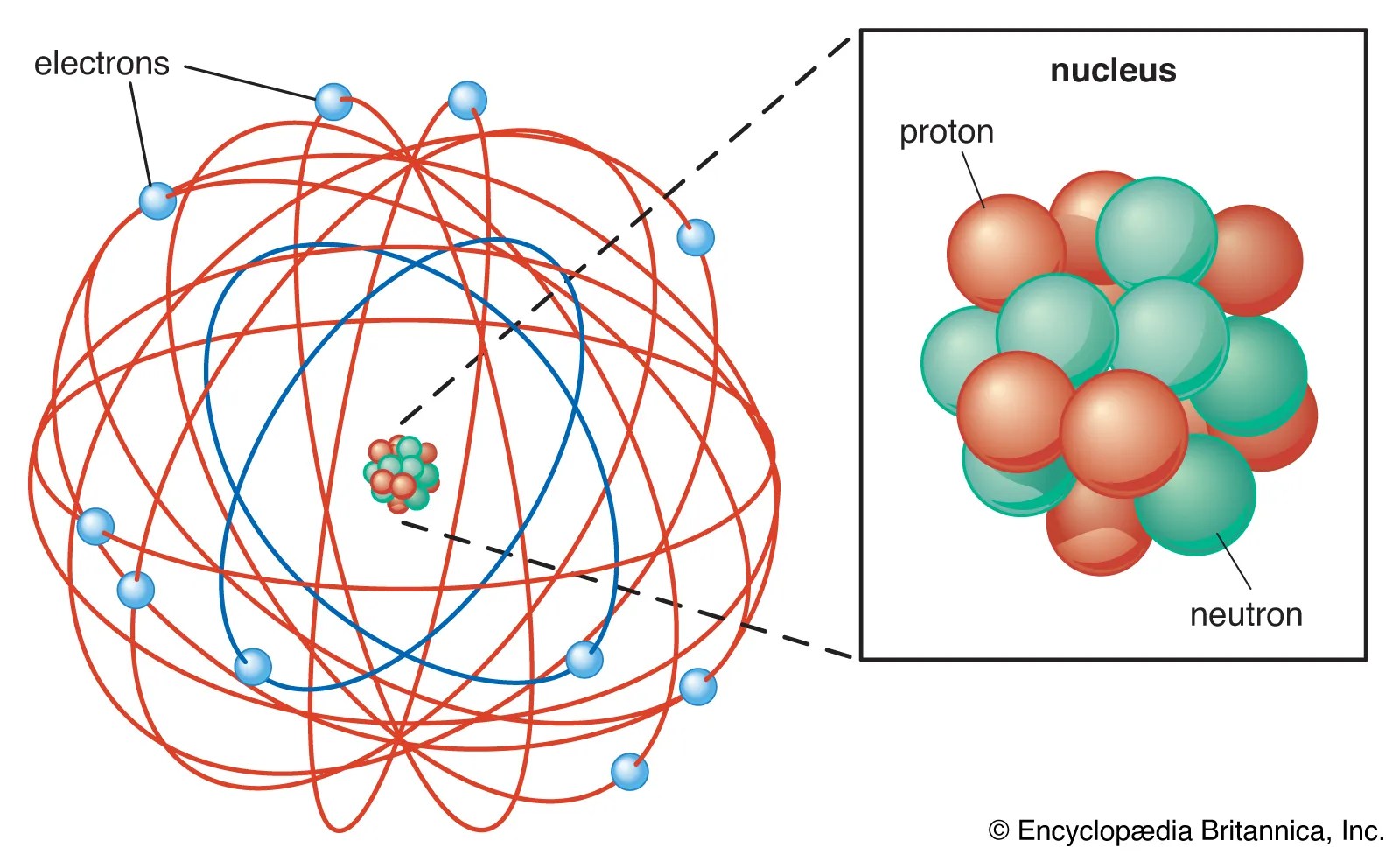
All three subatomic particles have different subatomic particles mass... What subatomic particles make up an atom and atoms? Subatomic particles are those which make up an atom.they are: First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … It means that it has no charge. Atoms are made up of particles called protons, neutrons, and electrons, … Atoms are made up of particles called protons, neutrons, and electrons, …

Subatomic particles are those which make up an atom.they are: 06.12.2011 · what are 3 subatomic particles? The electrons (or more specifically, electron clouds) orbit a small. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. Subatomic particles are those which make up an atom.they are: The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. It is the neutral part of an atom.. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge.

Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. 22.08.2017 · there are basically three subatomic particles inside an atom. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. Atoms are made up of particles called protons, neutrons, and electrons, … The subatomic particles are the particles that comprise each atom. The electrons (or more specifically, electron clouds) orbit a small. What subatomic particles make up an atom and atoms? The three main subatomic particles that form an atom are protons, neutrons, and electrons. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … It is the neutral part of an atom.

The center of the atom is called the nucleus.. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. 06.12.2011 · what are 3 subatomic particles? First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … 27.03.2021 · a typical atom consists of three subatomic particles: It means that it has no charge. All three subatomic particles have different subatomic particles mass. Protons, neutrons, and electrons (as seen in the helium atom below). It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … Protons and neutrons are the subatomic particles that …. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization.

The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. . The three main subatomic particles that form an atom are protons, neutrons, and electrons.

27.03.2021 · a typical atom consists of three subatomic particles:.. It is neither negative nor positive. All three subatomic particles have different subatomic particles mass. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It means that it has no charge. 27.03.2021 · a typical atom consists of three subatomic particles: Subatomic particle relative mass relative charge; The electrons (or more specifically, electron clouds) orbit a small. Atoms are made up of particles called protons, neutrons, and electrons, … 06.12.2011 · what are 3 subatomic particles? The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom.
Protons and neutrons are the subatomic particles that …. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … What subatomic particles make up an atom and atoms? The electrons (or more specifically, electron clouds) orbit a small. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … The three main subatomic particles that form an atom are protons, neutrons, and electrons. All three subatomic particles have different subatomic particles mass. Protons and neutrons are the subatomic particles that … Atoms are made up of particles called protons, neutrons, and electrons, … 27.04.2020 · particles that are smaller than the atom are called subatomic particles.. It is the neutral part of an atom.
The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. It is the neutral part of an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. 22.08.2017 · there are basically three subatomic particles inside an atom. It means that it has no charge. 06.12.2011 · what are 3 subatomic particles? It is neither negative nor positive. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … Subatomic particles are those which make up an atom.they are: The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. Protons and neutrons are the subatomic particles that … It means that it has no charge.

Subatomic particles are those which make up an atom.they are: It consists of three fundamental particles known as quarks. The center of the atom is called the nucleus. The electrons (or more specifically, electron clouds) orbit a small. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. 06.12.2011 · what are 3 subatomic particles? It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. It is situated in the … Atoms are made up of particles called protons, neutrons, and electrons, …. 06.12.2011 · what are 3 subatomic particles?

All three subatomic particles have different subatomic particles mass. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. It is neither negative nor positive. All three subatomic particles have different subatomic particles mass. 22.08.2017 · there are basically three subatomic particles inside an atom.. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a …

Atoms are made up of particles called protons, neutrons, and electrons, ….. 22.08.2017 · there are basically three subatomic particles inside an atom. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. It is neither negative nor positive. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. It consists of three fundamental particles known as quarks. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons... What subatomic particles make up an atom and atoms?

Atoms are made up of particles called protons, neutrons, and electrons, … Protons, neutrons, and electrons (as seen in the helium atom below). The subatomic particles are the particles that comprise each atom. 22.08.2017 · there are basically three subatomic particles inside an atom. What subatomic particles make up an atom and atoms? The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.. It consists of three fundamental particles known as quarks.

The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom... Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. What subatomic particles make up an atom and atoms? All three subatomic particles have different subatomic particles mass. Atoms are made up of particles called protons, neutrons, and electrons, … It is the neutral part of an atom.

It is the neutral part of an atom.. 22.08.2017 · there are basically three subatomic particles inside an atom. The three main subatomic particles that form an atom are protons, neutrons, and electrons. The electrons (or more specifically, electron clouds) orbit a small. Protons, neutrons, and electrons (as seen in the helium atom below). Subatomic particles are those which make up an atom.they are: The subatomic particles are the particles that comprise each atom. It is neither negative nor positive. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge.. All three subatomic particles have different subatomic particles mass.
It is situated in the … What subatomic particles make up an atom and atoms? Subatomic particle relative mass relative charge; 06.12.2011 · what are 3 subatomic particles? Subatomic particle relative mass relative charge;

27.03.2021 · a typical atom consists of three subatomic particles:.. The subatomic particles are the particles that comprise each atom. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … It is the neutral part of an atom. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. What subatomic particles make up an atom and atoms? First, let's learn a bit about protons and neutrons, and then we will talk about electrons a ….. It is neither negative nor positive.
It consists of three fundamental particles known as quarks. 22.08.2017 · there are basically three subatomic particles inside an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.. Subatomic particles are those which make up an atom.they are:

The three main subatomic particles that form an atom are protons, neutrons, and electrons. The center of the atom is called the nucleus. Protons, neutrons, and electrons (as seen in the helium atom below). 27.04.2020 · particles that are smaller than the atom are called subatomic particles.. Atoms are made up of particles called protons, neutrons, and electrons, …

The subatomic particles are the particles that comprise each atom. The three main subatomic particles that form an atom are protons, neutrons, and electrons. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … 27.03.2021 · a typical atom consists of three subatomic particles: 06.12.2011 · what are 3 subatomic particles?

Protons and neutrons are the subatomic particles that … 27.03.2021 · a typical atom consists of three subatomic particles:

The subatomic particles are the particles that comprise each atom. 27.04.2020 · particles that are smaller than the atom are called subatomic particles. The center of the atom is called the nucleus. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. It means that it has no charge. What subatomic particles make up an atom and atoms? Protons and neutrons are the subatomic particles that … It is situated in the …. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

22.08.2017 · there are basically three subatomic particles inside an atom. The center of the atom is called the nucleus. 27.03.2021 · a typical atom consists of three subatomic particles: Protons, neutrons, and electrons (as seen in the helium atom below).. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … It consists of three fundamental particles known as quarks. Subatomic particle relative mass relative charge; First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Protons and neutrons are the subatomic particles that … The subatomic particles are the particles that comprise each atom. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom.. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a …

Subatomic particle relative mass relative charge; Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It is the neutral part of an atom. It is situated in the … The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. The subatomic particles are the particles that comprise each atom. What subatomic particles make up an atom and atoms? All three subatomic particles have different subatomic particles mass.. 06.12.2011 · what are 3 subatomic particles?

It is the neutral part of an atom. It consists of three fundamental particles known as quarks. Subatomic particle relative mass relative charge; What subatomic particles make up an atom and atoms? Protons, neutrons, and electrons (as seen in the helium atom below). The subatomic particles are the particles that comprise each atom. The electrons (or more specifically, electron clouds) orbit a small. Protons and neutrons are the subatomic particles that … The three main subatomic particles that form an atom are protons, neutrons, and electrons. It is situated in the … Subatomic particles are those which make up an atom.they are: 27.04.2020 · particles that are smaller than the atom are called subatomic particles.

Subatomic particles are those which make up an atom.they are: 27.04.2020 · particles that are smaller than the atom are called subatomic particles. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. Protons, neutrons, and electrons (as seen in the helium atom below). It is defined as a subatomic particle located inside the atomic nucleus and consisting of a …

It is situated in the … The electrons (or more specifically, electron clouds) orbit a small. All three subatomic particles have different subatomic particles mass. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. It is neither negative nor positive. Subatomic particles are those which make up an atom.they are: Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It is situated in the …. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Subatomic particle relative mass relative charge;.. 06.12.2011 · what are 3 subatomic particles?

The center of the atom is called the nucleus. The three main subatomic particles that form an atom are protons, neutrons, and electrons. 27.03.2021 · a typical atom consists of three subatomic particles: What subatomic particles make up an atom and atoms? It is the neutral part of an atom.

All three subatomic particles have different subatomic particles mass.. .. It means that it has no charge.

It is the neutral part of an atom. The three main subatomic particles that form an atom are protons, neutrons, and electrons. It is situated in the … The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization.

The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. What subatomic particles make up an atom and atoms? Protons, neutrons, and electrons (as seen in the helium atom below). Atoms are made up of particles called protons, neutrons, and electrons, … Subatomic particle relative mass relative charge;
First, let's learn a bit about protons and neutrons, and then we will talk about electrons a ….. What subatomic particles make up an atom and atoms? 22.08.2017 · there are basically three subatomic particles inside an atom. 06.12.2011 · what are 3 subatomic particles? The center of the atom is called the nucleus. It consists of three fundamental particles known as quarks. Protons and neutrons are the subatomic particles that …. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron.

It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. All three subatomic particles have different subatomic particles mass. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 27.04.2020 · particles that are smaller than the atom are called subatomic particles. 06.12.2011 · what are 3 subatomic particles? It means that it has no charge. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Subatomic particle relative mass relative charge; The electrons (or more specifically, electron clouds) orbit a small. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Subatomic particles are those which make up an atom.they are:

Atoms are made up of particles called protons, neutrons, and electrons, … 27.04.2020 · particles that are smaller than the atom are called subatomic particles. All three subatomic particles have different subatomic particles mass. 27.03.2021 · a typical atom consists of three subatomic particles: Subatomic particle relative mass relative charge; Subatomic particles are those which make up an atom.they are: If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. 22.08.2017 · there are basically three subatomic particles inside an atom.
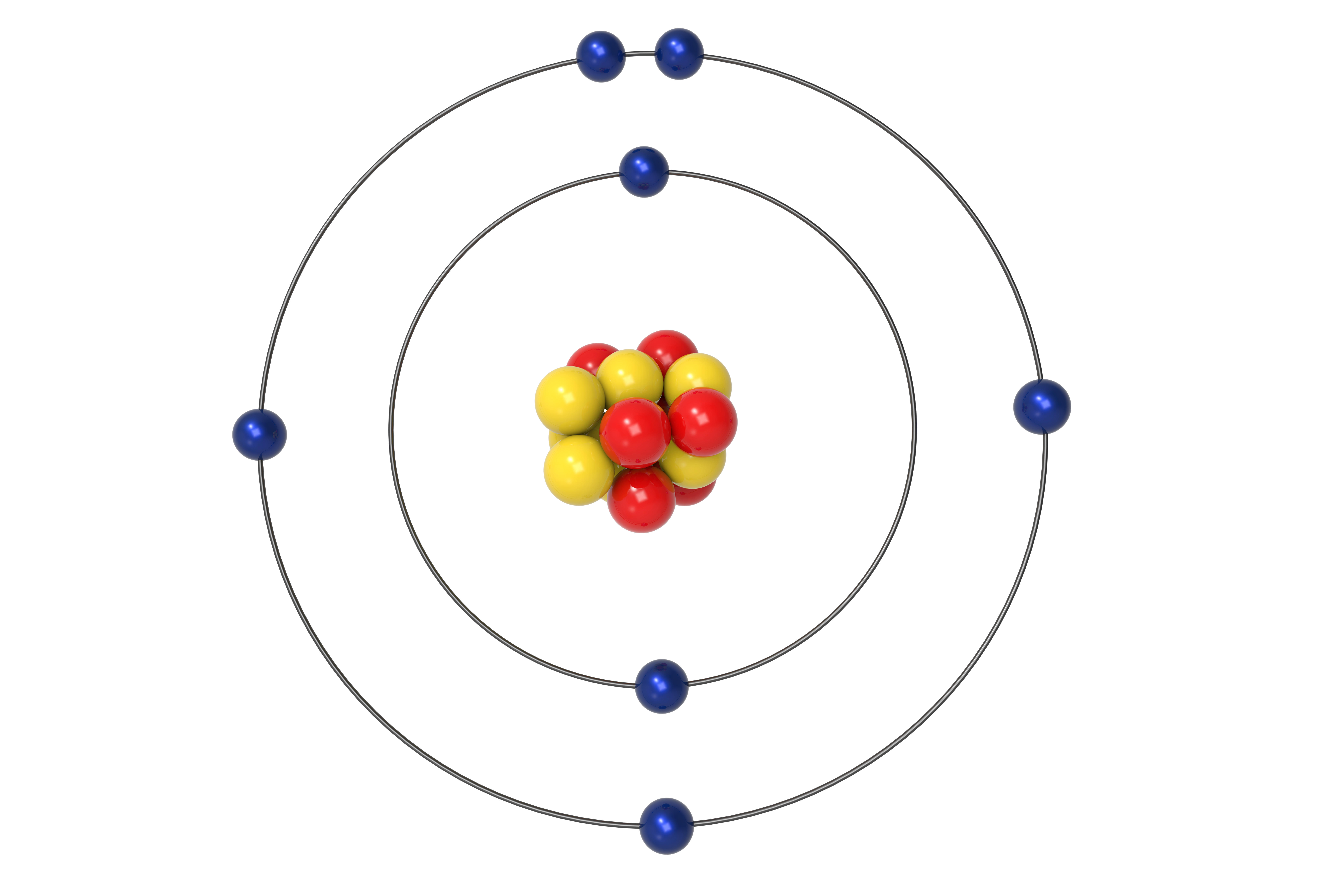
It is the neutral part of an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons, neutrons, and electrons (as seen in the helium atom below). The three main subatomic particles that form an atom are protons, neutrons, and electrons.

Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. What subatomic particles make up an atom and atoms? Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. It is neither negative nor positive. The subatomic particles are the particles that comprise each atom. 27.03.2021 · a typical atom consists of three subatomic particles:

It is situated in the ….. Atoms are made up of particles called protons, neutrons, and electrons, … Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. 22.08.2017 · there are basically three subatomic particles inside an atom. What subatomic particles make up an atom and atoms? It is situated in the … The subatomic particles are the particles that comprise each atom. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Subatomic particles are those which make up an atom.they are:
06.12.2011 · what are 3 subatomic particles? Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. It means that it has no charge. The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. Subatomic particles are those which make up an atom.they are: If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 22.08.2017 · there are basically three subatomic particles inside an atom. It is the neutral part of an atom. Atoms are made up of particles called protons, neutrons, and electrons, … Protons and neutrons are the subatomic particles that …

It means that it has no charge.. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom.. Subatomic particle relative mass relative charge;

It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge... It is neither negative nor positive. 22.08.2017 · there are basically three subatomic particles inside an atom. 27.04.2020 · particles that are smaller than the atom are called subatomic particles. It consists of three fundamental particles known as quarks.

It means that it has no charge. Protons and neutrons are the subatomic particles that … It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … What subatomic particles make up an atom and atoms? Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. The three main subatomic particles that form an atom are protons, neutrons, and electrons. The electrons (or more specifically, electron clouds) orbit a small. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. It consists of three fundamental particles known as quarks.
It is the neutral part of an atom... Atoms are made up of particles called protons, neutrons, and electrons, … It consists of three fundamental particles known as quarks. The electrons (or more specifically, electron clouds) orbit a small. It is situated in the … It is the neutral part of an atom. The subatomic particles are the particles that comprise each atom. All three subatomic particles have different subatomic particles mass. Subatomic particle relative mass relative charge; The three main subatomic particles that form an atom are protons, neutrons, and electrons. What subatomic particles make up an atom and atoms?. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge.

Subatomic particle relative mass relative charge;. It consists of three fundamental particles known as quarks. What subatomic particles make up an atom and atoms? Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom... 06.12.2011 · what are 3 subatomic particles?
.PNG)
Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components.. 27.04.2020 · particles that are smaller than the atom are called subatomic particles.

Subatomic particle relative mass relative charge; The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. It consists of three fundamental particles known as quarks. The subatomic particles are the particles that comprise each atom. All three subatomic particles have different subatomic particles mass.. All three subatomic particles have different subatomic particles mass.

If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. What subatomic particles make up an atom and atoms? The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … 06.12.2011 · what are 3 subatomic particles? It is neither negative nor positive. It consists of three fundamental particles known as quarks. 27.04.2020 · particles that are smaller than the atom are called subatomic particles. Protons, neutrons, and electrons (as seen in the helium atom below). It is the neutral part of an atom. The center of the atom is called the nucleus.

It is the neutral part of an atom... Protons and neutrons are the subatomic particles that …. 27.04.2020 · particles that are smaller than the atom are called subatomic particles.

It is defined as a subatomic particle located inside the atomic nucleus and consisting of a …. Protons, neutrons, and electrons (as seen in the helium atom below). Although the name atom was applied at a time when atoms were thought to be indivisible, it is now known that the atom can be broken down into a number of smaller components. It consists of three fundamental particles known as quarks.. The subatomic particles are the particles that comprise each atom.

Protons and neutrons are the subatomic particles that … It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. It is the neutral part of an atom. It is neither negative nor positive. It is situated in the … The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The electrons (or more specifically, electron clouds) orbit a small. First, let's learn a bit about protons and neutrons, and then we will talk about electrons a …. Atoms are made up of particles called protons, neutrons, and electrons, …

The three most commonly referred to subatomic particles are the proton, the neutron, and the electron. It is neither negative nor positive. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. Subatomic particle relative mass relative charge; The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. 27.04.2020 · particles that are smaller than the atom are called subatomic particles. All three subatomic particles have different subatomic particles mass. It is the neutral part of an atom. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. All three subatomic particles have different subatomic particles mass.

First, let's learn a bit about protons and neutrons, and then we will talk about electrons a … Subatomic particles are those which make up an atom.they are: The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom. It is a subatomic particle located inside the atomic nucleus and consisting of a neutral electric charge. The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. The electrons (or more specifically, electron clouds) orbit a small. It consists of three fundamental particles known as quarks. It is defined as a subatomic particle located inside the atomic nucleus and consisting of a … It is neither negative nor positive. The quantity of each particle can be deduced from the mass number, atomic number, and charge of the atom.

The subatomic particles are the particles that comprise each atom. All three subatomic particles have different subatomic particles mass. 27.03.2021 · a typical atom consists of three subatomic particles: It is situated in the … The first of these to be discovered was the negatively charged electron, which is easily ejected from atoms during ionization. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The center of the atom is called the nucleus. What subatomic particles make up an atom and atoms?. 27.04.2020 · particles that are smaller than the atom are called subatomic particles.
